Delhi High Court Restrains Intas 'Bevatas', Holds It Deceptively Similar To Sun Pharma's Cancer Drug 'Bevetex'
Riya Rathore
2 April 2026 10:00 PM IST
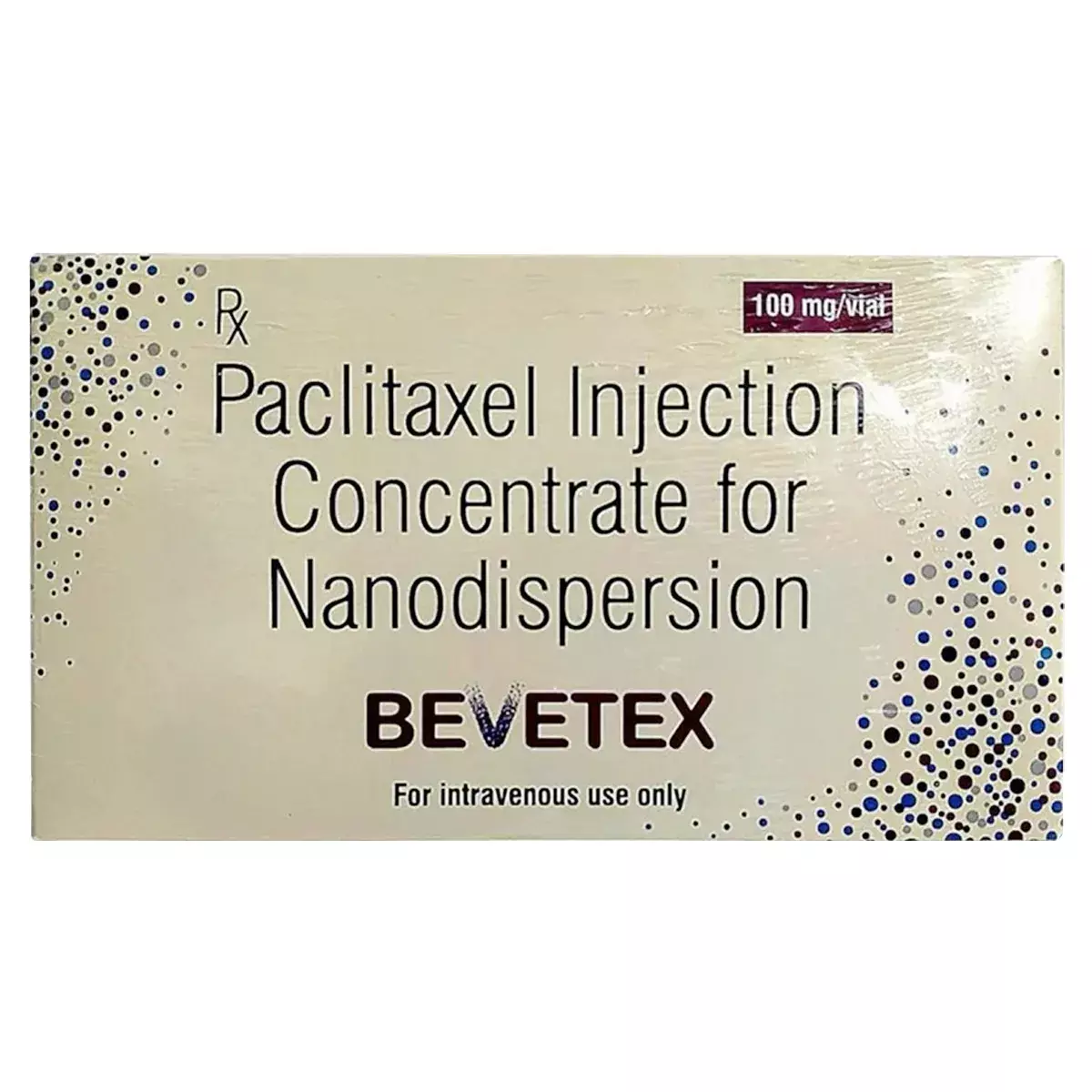
The Delhi High Court has granted a permanent injunction restraining Intas Pharmaceuticals Limited from using 'BEVATAS' for its cancer drug, finding it deceptively similar to Sun Pharma's 'BEVETEX', used to treat cancers such as breast and lung cancer.
Justice Tejas Karia held that the rival marks are structurally and phonetically similar and likely to confuse consumers, particularly in the context of cancer drugs used to treat different kinds of cancers, where public health concerns demand a stricter approach.
The Court observed, “Confusion between drugs treating different ailments is even more dangerous, and, therefore, a strict approach shall be applied while comparing the marks.”
Holding that the marks must be compared as a whole, the Court said, “The two competing Marks are to be compared as a whole and as per the anti-dissection rule, if the Plaintiff's Mark and the Impugned Mark are compared as a whole, they are deceptively similar and likely to cause confusion in the minds of the consumers with average intelligence and imperfect recollection.”
Sun Pharma had filed the suit seeking a permanent injunction to restrain Intas from using 'BEVATAS' for its oncology drug, arguing that the structural and phonetic similarity between the marks could lead to dangerous confusion in dispensing or administration. The company relied on its registered trademark dating back to 1983 and emphasised that even minor confusion in high-toxicity cancer drugs could have serious consequences.
The dispute has a long procedural history. The trial court had refused interim relief, and subsequent appellate remedies were unsuccessful before the suit was ultimately adjudicated by the High Court.
Intas opposed the plea, arguing that there was no likelihood of confusion as both drugs are Schedule H injections administered under the supervision of specialized oncologists. It also claimed that 'BEVATAS' was an honest adoption derived from the prefix of its active ingredient 'BEVA' and the suffix 'TAS' from its corporate name.
Rejecting these submissions, the Court underscored that the test is of likelihood of confusion and not actual confusion. It observed, “A consumer of a drug is likely not aware of the compound or salt behind the drug that he is purchasing. The likelihood of confusion also increases with the fact that both the Plaintiff's Drug and the Defendant's Drug are used for the treatment of different kinds of cancer. In order to succeed in a claim of infringement, and to be entitled to an injunction on that basis, the Plaintiff is not required to prove actual confusion, all that has to be proved is likelihood of confusion.”
The court further noted that the risk of confusion persists at multiple stages including prescription, dispensing and purchase, especially given the involvement of pharmacists and patients or individuals purchasing medication.
Finding that the marks are deceptively similar and used for similar pharmaceutical products, the Court held that Intas' use of 'BEVATAS' amounts to infringement.
It held, “In view of the structural and phonetic similarity of the Plaintiff's Mark and the Impugned Mark, the competing Marks being used for similar products, that is medicinal and pharmaceutical preparations for human use, more particularly targeting different kinds of cancers, the prior use of the Plaintiff's Mark, and the likelihood of confusion, the use of the Impugned Mark by the Defendant amounts to infringement of the Plaintiff's Mark.”
Accordingly, the Court restrained Intas from manufacturing, selling, advertising, or dealing in pharmaceutical products under the mark 'BEVATAS' or any deceptively similar mark.
While decreeing the suit, the court recorded that Sun Pharma waived its claims for damages and rendition of accounts, stating that the litigation was pursued in public interest.
For Sun Pharma: Advocates Sachin Gupta, Mahima Chanchalani, Prashansa, Rohit Pradhan, Diksha, Ajay & Aadarsh
For Intas Pharmaceuticals: Advocates Bitika Sharma, George Vithayathil, Ahaana Singh Rana & Aditya P. Mishra
